TruSight Oncology 500 Portfolio
TruSight Oncology 500 Assay
Now with HRD* *Not available in Japan
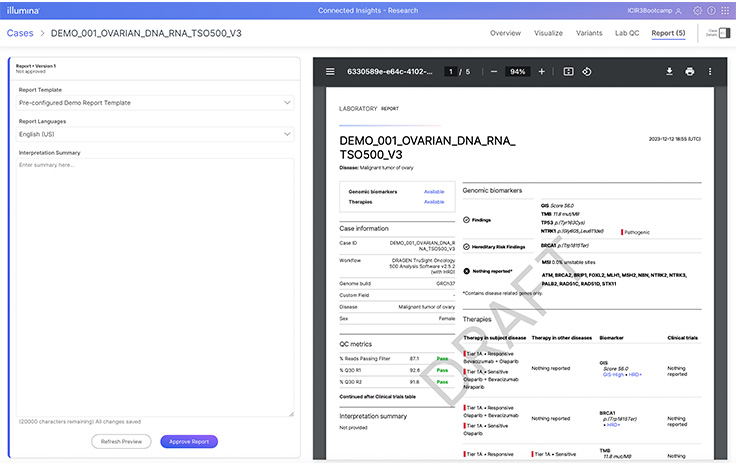
Assess relevant DNA and RNA cancer biomarkers from FFPE tumor tissue, now including HRD through an accessory kit to assess the Genomic Instability Score (GIS).

TruSight Oncology 500 High-Throughput Assay
Batch up to 192 samples at a time while using the same panel content and tissue input type as the TruSight Oncology 500 assay.
TruSight Oncology 500 ctDNA v2 Assay
Analyze circulating tumor DNA (ctDNA) from blood plasma with a more sensitive and streamlined assay in <4 days with automation-friendly options.
Benefits of TruSight Oncology 500 Portfolio
Enable Comprehensive Genomic Profiling
Bring the Assay into Your Lab
Integrated, Flexible, and Scalable Workflow
Achieve Timely and Reliable Results

TruSight Oncology 500 Portfolio Brochure
This brochure provides an overview of the TSO 500 product portfolio. You'll learn about Its broad coverage, integrated workflows, automation capabilities, easy-to-use data analysis and more. Take cancer from uncertainty to insight with streamlined solutions enabling high-quality cmprehensive genomic profiling.
Download TSO 500 Portfolio Brochure†NovaSeq 6000Dx System in RUO Mode requires a separate, stand-alone DRAGEN server for secondary analysis.
‡Coming soon in 2024.
Pan-Cancer Biomarkers
Detection of currently relevant DNA & RNA* variants for multiple types of solid tumors. Some key biomarkers include:
| Pan-cancer: BRAFNTRK1 NTRK2 NTRK3RET MSI TMB | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Genes with biomarkers of significance† | Genes with biomarkers of potential significance‡ | ||||||||||
 Breast Breast |
BRCA1 | BRCA2 | ERBB2 | ESR1 | PALB2 | PIK3CA | 180 | ||||
 Colorectal Colorectal |
ERBB2 | KRAS | NRAS | 166 | |||||||
 Bone Bone |
EGFR | ERG | ETV1 | ETV4 | EWSR1 | FEV | FLI1 | FUS | H3F3A | 140 | |
| HEY1 | IDH1 | MDM2 | NCOA2 | SMARCB1 | |||||||
 Lung Lung |
ALK | EGFR | ERBB2 | KRAS | MET | NUTM1 | ROS1 | 223 | |||
 Melanoma Melanoma |
KIT | NRAS | ROS1 | 172 | |||||||
 Ovarian Ovarian |
BRCA1 | BRCA2 | FOXL2 | 149 | |||||||
 CNS CNS |
APC | ATRX | CDKN2A | CDKN2B | EGFR | H3F3A | HIST1H3B | HIST1H3C | IDH1 | 140 | |
| IDH2 | MYCN | PTCH1 | RELA | TERT | TP53 | ||||||
 Prostate Prostate |
AR | ATM | BARD1 | BRCA1 | BRCA2 | BRIP1 | CDK12 | CHEK1 | CHEK2 | 151 | |
| FANCL | FGFR2 | FGFR3 | PALB2 | RAD51B | RAD51C | RAD51D | RAD54L | ||||
 Thyroid Thyroid |
HRAS | KRAS | NRAS | RET | TERT | 165 | |||||
 Uterine & cervical Uterine & cervical |
BRCA2 | EPC1 | ERBB2 | ESR1 | FOXO1 | GREB1 | JAZF1 | NCOA2 | NCOA3 | 138 | |
| NUTM2A | NUTM2B | PAX3 | PAX7 | PHF1 | POLE | SMARCA4 | SUZ12 | TP53 | |||
| YWHAE | |||||||||||
 Other Solid Tumors Other Solid Tumors |
ALK | APC | ARID1A | ASPSCR1 | ATF1 | ATIC | BAP1 | BCOR | BRCA1 | 152 | |
| BRCA2 | CAMTA1 | CARS | CCNB2 | CDK4 | CDKN2A | CIC | CITED2 | CLTC | |||
| COL1A1 | COL6A3 | CREB1 | CREB3L1 | CREB3L2 | CSF1 | CTNNB1 | DDIT3 | DDX3X | |||
| DNAJB1 | DUX4 | EED | EGFR | ERBB2 | ERG | ETV1 | ETV4 | ETV6 | |||
| EWSR1 | FEV | FGFR2 | FGFR3 | FLI1 | FOXL2 | FOXO1 | FOXO4 | FUS | |||
| GLI1 | HEY1 | HGF | HMGA2 | IDH1 | KRAS | LEUTX | MAML3 | MDM2 | |||
| MYB | MYOD1 | NAB2 | NCOA2 | NF1 | NFATC2 | NFIB | NR4A3 | NRAS | |||
| NUTMI | NUTM2A | NUTM2B | PALB2 | PATZ1 | PAX3 | PAX7 | PDGFB | PDGFRA | |||
| PRKACA | PRKD1 | RANBP2 | ROS1 | SDHA | SDHB | SDHC | SDHD | SMARCB1 | |||
| SS18 | SSX1 | SSX2 | SSX4 | STAT6 | SUZ12 | TAF15 | TCF12 | TERT | |||
| TFE3 | TFEB | TFG | TP53 | TPM3 | TPM4 | TRAF7 | TSPAN31 | VGLL2 | |||
| WT1 | WWTR1 | YAP1 | YWHAE | ZC3H7B | |||||||
The genes and biomarkers listed in this table are a subset of all genes included in the panel. To see the full gene list, view the product datasheet, available under Product Literature on the respective product pages.
* RNA variants only called from TSO 500 and TSO 500 HT assays.
† The product to evaluate DNA & RNA variants is the TruSight Oncology 500 DNA/RNA Bundle.

Analytical Evaluation & Verification Service
The TruSight Oncology 500 Analytical Evaluation Service facilitates alignment with the latest CAP, AMP, and European standards. Use the service to evaluate analytical performance characteristics and integrate products into your workflows while reducing overall evaluation time by ~60%.
View Service